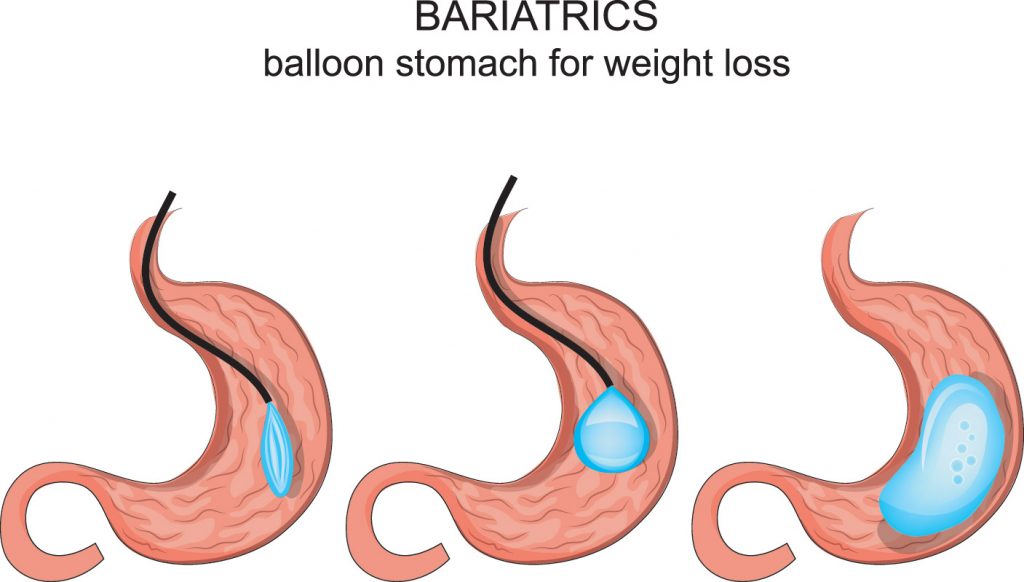
The gastric balloon procedure is a non-surgical alternative to weight loss surgery, such as gastric bypass. Unlike weight loss surgery, the balloon procedure is temporary, with the balloon being removed after six months. It does not permanently alter your digestive system. It is also considered to be a low-risk procedure. But, the U.S. Food and Drug Administration (FDA) has recently warned health care professionals of seven deaths associated with liquid-filled intragastric balloon systems. The cause of the problem is not yet known.
The FDA Warning
On August 10, 2017, the FDA issued a letter to healthcare professional warning of five reports of unexpected deaths in patients who had received liquid-filled intragastric balloon systems for treatment of obesity. All five reports showed death occurring within a month of balloon placement, with three reports of the death occurring within one to three days of placement.
One report the ReShape Integrated Dual Balloon System, manufactured by ReShape Medical Inc, with the remaining four involving the Orbera Intragastric Balloon System, manufactured by Apollo Endo Surgery.
The FDA does not know what caused the deaths in those five reports, but noted that there were two additional reports of death during the same time period for which the potential cause was known. One involved gastric perforation with the Orbera Intragastric Balloon System and the other involved esophageal perforation in a procedure using the ReShape Integrated Dual Balloon System.
If you believe that you or a loved one has been harmed by a gastric balloon, please talk to an experienced defective drug attorney right away to learn more about your rights and potential compensation.
Leave a Reply