Summary Company Announcement Date: February 13, 2025 FDA Publish Date: February 14, 2025 Product Type: Drugs … [Read more...]
Search Results for: labeling
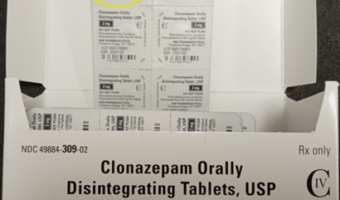
Endo Expands Voluntary Recall of Clonazepam Orally Disintegrating Tablets, USP (C-IV) Due to Potential Product Carton Strength Mislabeling
Summary Company Announcement Date: November 18, 2024 FDA Publish Date: November 19, 2024 Product Type: Drugs … [Read more...]
Endo USA, Inc. Issues Voluntary, Nationwide Recall of One Lot of Clonazepam Orally Disintegrating Tablets, USP (C-IV) Lot Number 550147301 Due to Mislabeling: Incorrect Strength on Product Carton
Summary Company Announcement Date: July 16, 2024 FDA Publish Date: July 17, 2024 Product Type: Drugs … [Read more...]
Compare Brands Recalls ADIOS! Super Vinegar All Natural Cleaner Due to Risk of Poisoning and Chemical Burns; Violation of Labeling Requirements under the Federal Hazardous Substances Act; Sold Exclusively on Amazon.com
The recalled cleaning product violates the Federal Hazardous Substances Act because it is deceptively labeled as “Safe to Use” and lacks the required cautionary statements for a poison, posing a risk of poisoning and chemical burns to consumers. … [Read more...]
ResMed Notifies Customers About Updated Instructions and Labeling for Masks with Magnets Due to Potential Interference with Certain Medical Devices
Summary Company Announcement Date: December 20, 2023 FDA Publish Date: December 21, 2023 Product Type: Medical Devices … [Read more...]



Recent Comments