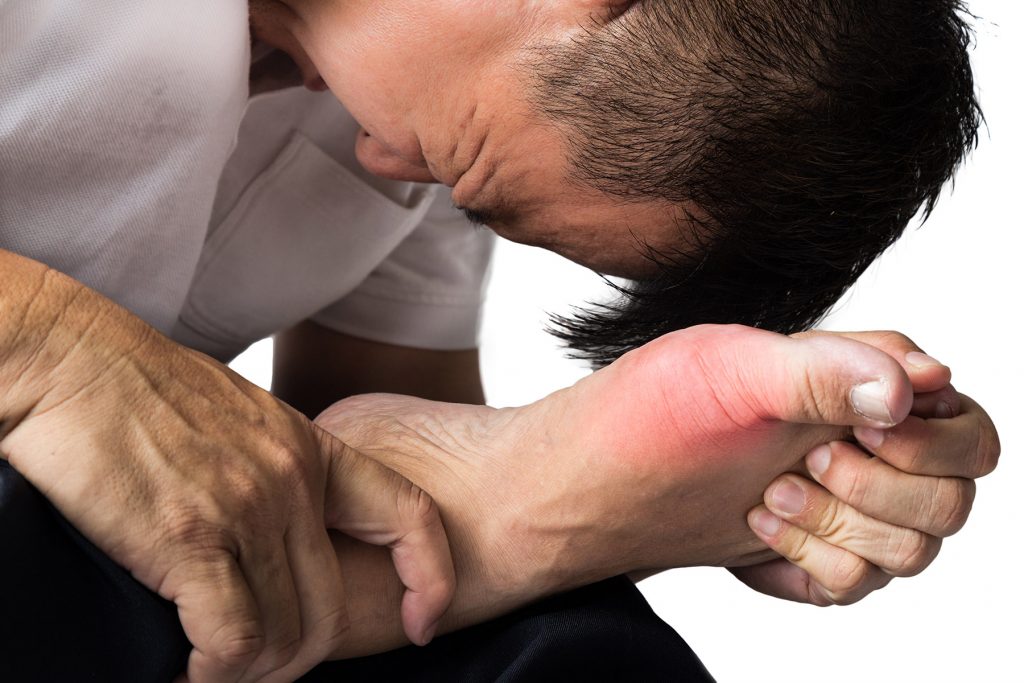
The Food and Drug Administration (FDA) posted an urgent message on its website on February 21, 2019: “The FDA has concluded there is an increased risk of death with Uloric (febuxostat) compared to another gout medicine, allopurinol. This conclusion is based on our in-depth review of results from a safety clinical trial that found an increased risk of heart-related death and death from all causes with Uloric.”
According to the FDA, people taking Uloric for the treatment of gout may face an increased risk of:
- Cardiac event-related death
- Non-deadly heart attack
- Non-deadly stroke
- Unstable angina
- Death
The FDA has also added a black box warning to the Uloric label (black box warnings are the strongest ones placed on prescription medication):
“Gout patients with established cardiovascular (CV) disease treated with Uloric had a higher rate of CV death compared to those treated with allopurinol in a CV outcomes study. Consider the risks and benefits of Uloric when deciding to prescribe or continue patients on Uloric. Uloric should only be used in patients who have an inadequate response to a maximally titrated dose of allopurinol, who are intolerant to allopurinol, or for whom treatment with allopurinol is not advisable.”
Allopurinol has been used to treat gout for decades, is safe, and effective notes the FDA.
Research has raised serious concerns about febuxostat’s cardiac risk. In its February announcement, the FDA pointed to the findings of a postmarket clinical trial of 6,190 patients with gout who were treated with febuxostat and allopurinol. “In patients treated with Uloric, 15 deaths from heart-related causes were observed for every 1,000 patients treated for a year compared to 11 deaths from heart-related causes per 1,000 patients treated with allopurinol for a year.”
Simply put, the FDA says there’s an increased risk of death associated with Uloric when compared to the popular gout medication, allopurinol. Uloric has shown an increased risk of heart-related death, as well as death from other causes associated with the drug.
Whistleblower Exposes Drug Company’s Questionable Actions
Takeda Pharmaceuticals, the maker of Uloric, has been accused of suppressing information about the serious side effects of Uloric, which was approved by the FDA under the condition that the “manufacturer conduct post-marketing clinical trials to compare the rates of cardiovascular events linked to Uloric and allopurinol.”
That research was never done, and a former Takeda Pharmaceuticals employee filed a whistleblower lawsuit that alleged the drug maker was withholding crucial information about the side effects of Uloric, including a risk of death when the gout drug was used with medications digoxin and the popular blood thinner, warfarin. The whistleblower also claimed that Takeda “altered and falsified adverse event reports to hide potential side effects, which could include bone marrow failure, severe bleeding, and kidney failure.” This was done, according to the lawsuit, in an effort to make Uloric a viable competitor against allopurinol.
If you’re currently taking Uloric, please contact your doctor to find out if you’re at risk of any of the side effects listed here. Please do not stop the drug without first speaking to your physician as some drugs cannot be stopped abruptly. People with any of these symptoms who are taking the drug, Uloric (generic febuxostat) should call their doctor:
- Chest pain
- Difficulty breathing
- Rapid or irregular heart rate
- Numbness or weakness affecting one side of the
- Dizziness, vertigo, or trouble standing
- Difficulty speaking (forming sentences)
- Severe headache that comes on quickly
Leave a Reply